|
12/9/2023 0 Comments Vsepr shapes linearThe electron-pair geometries will be the same as the molecular structures when there are no lone electron pairs around the central atom, but they will be different when there are lone pairs present on the central atom. The structure that includes only the placement of the atoms in the molecule is called the molecular structure. We differentiate between these two situations by naming the geometry that includes all electron pairs the electron-pair geometry. The molecular structure of the methane molecule, CH 4, is shown with a tetrahedral arrangement of the hydrogen atoms. Two regions of electron density around a central atom in a molecule form a linear geometry three regions form a trigonal planar geometry four regions form a tetrahedral geometry five regions form a trigonal bipyramidal geometry and six regions form an octahedral geometry.įigure 4. Figure 3 illustrates this and other electron-pair geometries that minimize the repulsions among regions of high electron density (bonds and/or lone pairs). With two bonds and no lone pairs of electrons on the central atom, the bonds are as far apart as possible, and the electrostatic repulsion between these regions of high electron density is reduced to a minimum when they are on opposite sides of the central atom. The Lewis structure of CO 2 (Figure 2) shows only two electron pairs around the central carbon atom. Other interactions, such as nuclear-nuclear repulsions and nuclear-electron attractions, are also involved in the final arrangement that atoms adopt in a particular molecular structure.Īs a simple example of VSEPR theory, let us predict the structure of a gaseous CO 2 molecule. We should understand, however, that the theory only considers electron-pair repulsions. VSEPR theory predicts the arrangement of electron pairs around each central atom and, usually, the correct arrangement of atoms in a molecule. The electrostatic repulsion of these electrons is reduced when the various regions of high electron density assume positions as far from each other as possible. The electrons in the valence shell of a central atom form either bonding pairs of electrons, located primarily between bonded atoms, or lone pairs. The VSEPR model assumes that electron pairs in the valence shell of a central atom will adopt an arrangement that minimizes repulsions between these electron pairs by maximizing the distance between them. Valence shell electron-pair repulsion theory (VSEPR theory) enables us to predict the molecular structure, including approximate bond angles around a central atom, of a molecule from an examination of the number of bonds and lone electron pairs in its Lewis structure. Bond distances (lengths) and angles are shown for the formaldehyde molecule, H 2CO. 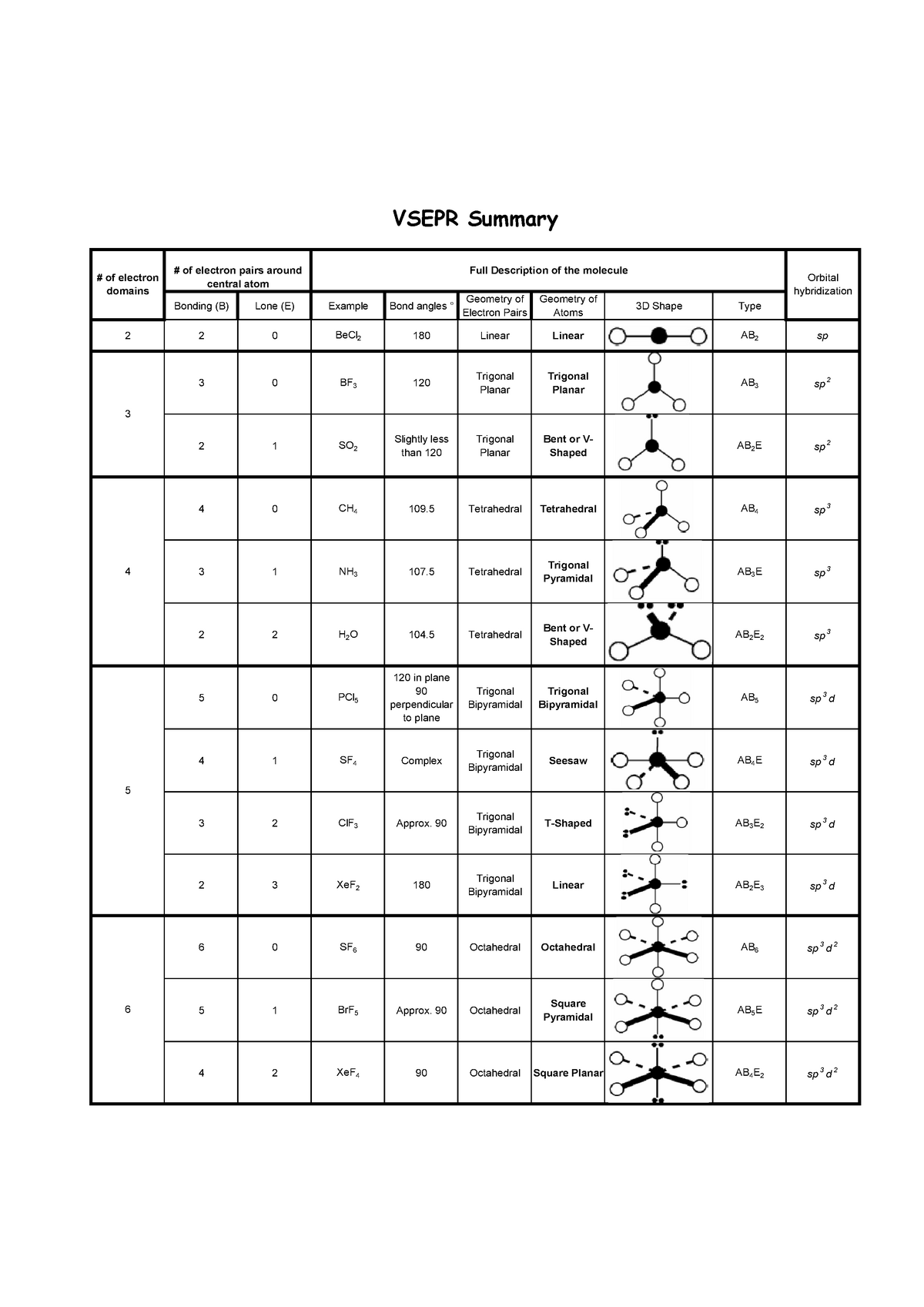
As described by the VSEPR model, the five valence electron pairs on the central atom form a trigonal bipyramid in which the three lone pairs occupy the less crowded equatorial positions and the two bonded atoms occupy the two axial positions at the opposite ends of an axis, forming a linear molecule.Figure 1. Linear geometry also occurs in AX 2E 3 molecules, such as xenon difluoride ( XeF 2) and the triiodide ion ( I − 3) with one iodide bonded to the two others. Linear anions include azide ( N −=N +=N −) and thiocyanate ( S=C=N −), and a linear cation is the nitronium ion ( O=N +=O). The most important linear molecule with more than three atoms is acetylene ( H−C≡C−H), in which each of its carbon atoms is considered to be a central atom with a single bond to one hydrogen and a triple bond to the other carbon atom. Neutral AX 2 molecules with linear geometry include beryllium fluoride ( F−Be−F) with two single bonds, carbon dioxide ( O=C=O) with two double bonds, hydrogen cyanide ( H−C≡N) with one single and one triple bond. Linear organic molecules, such as acetylene ( HC≡CH), are often described by invoking sp orbital hybridization for their carbon centers.Īccording to the VSEPR model (Valence Shell Electron Pair Repulsion model), linear geometry occurs at central atoms with two bonded atoms and zero or three lone pairs ( AX 2 or AX 2E 3) in the AXE notation. 
The linear molecular geometry describes the geometry around a central atom bonded to two other atoms (or ligands) placed at a bond angle of 180°. 
Structure of beryllium fluoride ( BeF 2), a compound with a linear geometry at the beryllium atom. 3D shape of molecules in which all bond angles are 180° Linear molecular geometry
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
